|
4/12/2023 0 Comments Cathode ray experiment conclusions 
Through experiments on evacuated tubes, Goldstein found that cathode rays could cast sharp shadows of emission perpendicular to the area covered by the cathode. Likewise, Eugen Godlstein's essays also contributed significantly to deepening technical notions about cathode rays. Hence, this type of phenomenon is known in the scientific world as channel rays, since they pass through the existing perforation in the cathode of the study tube. The positively charged ions that make up the channel rays move towards the perforated cathode until they pass through it, given the nature of the experiment. Goldstein named these rays "Kanalstrahlen" to refer to the counterpart of cathode rays. These cations are attracted to the negative electrode of the tube, given the natural attraction between opposite electrical charges. The gas atoms lose their negative charge, and become positively charged. Starting from this base, the electrons of the cathode ray repel the electrons of the gas atoms, and the latter are released from their original formation. 
Particles with equal charges repel each other. Channel raysĬhannel rays arise when cathode rays collide with atoms in the gas that is confined within the test tube. That is, the channel rays were positively charged. Because the channel rays traveled in the opposite direction to the cathode rays, Goldstein inferred that the nature of their electrical charge must also be opposite. This new flow was baptized by Goldstein as channel rays. In addition, the behavior of the particles in regard to their electric field and magnetic field, was totally opposite to that of cathode rays. Goldstein concluded that, in addition to the cathode rays, which traveled from the cathode (negative charge) towards the anode (positive charge), there was another ray that traveled in the opposite direction, that is, from the anode towards the cathode of the modified tube. However, the highlight is that these rays were moving in the opposite direction to the cathode rays and were called channel rays. Under this new configuration, Goldstein discovered that the tube emitted a new glow originating from the end of the tube that had been pierced. In addition, he repeated the experiment with the modification of the Crookes tube, increasing the voltage between the ends of the tube to several thousand volts. Goldstein modified the structure of the Crookes tube, adding several perforations to one of the metal cathodes in the tube. 
However, Ernest Rutherford is considered the discoverer in the scientific community. On the other hand, he is sometimes credited as the discoverer of the proton, which he observed in the vacuum tubes where he observed cathode rays. Godlstein did not propose an atomic model, although his discoveries allowed the development of Thomson's atomic model. One of his most outstanding legacies consisted in the discovery of what today are known as protons, together with channel rays, also known as anodic or positive rays. This discovery was made through experimentation with cathode ray tubes, in 1886. Goldstein identified the existence of protons as equal and opposite charges to electrons. His scientific work includes experiments with electrical phenomena in gases and cathode rays. First steps in the discovery of the protonĮugen goldstein He was a prominent German physicist, born in present-day Poland in 1850.ACTION OF HIGH SPEED ELECTRONS ON METHANE, OXYGEN AND CARBON MONOXIDE. THE ACTION OF HIGH-SPEED CATHODE RAYS ON THE SIMPLER ALCOHOLS, ALDEHYDES AND KETONES, AND ON ETHYLENE. Ozone hazards incurred by electron Van de Graaff operation. Human Factors: The Journal of the Human Factors and Ergonomics Society 1963, 5 Space Cabin Atmosphere Trace Contaminants and Their Possible Influence on Visual Parameters. Journal of Chemical & Engineering Data 2001, 46 Potentiometric Study of the Dissociation Quotients of Aqueous Dimethylammonium Ion As a Function of Temperature and Ionic Strength. Journal of Chemical & Engineering Data 2003, 48 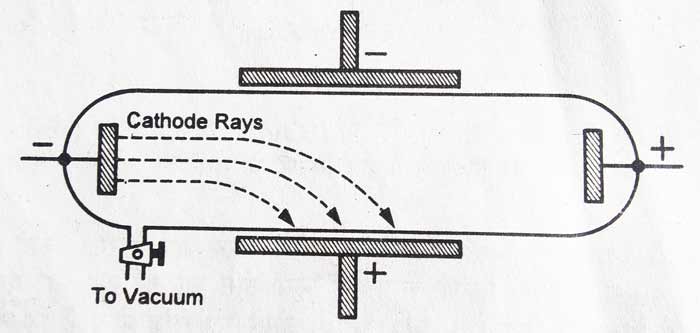
Determination of Stoichiometric Dissociation Constants of Acetic Acid in Aqueous Solutions Containing Acetic Acid, Sodium Acetate, and Sodium Chloride at (0 to 60) ☌. Journal of Chemical & Engineering Data 2005, 50 Re-evaluation of Stoichiometric Dissociation Constants from Electrochemical Cell Data for Formic Acid at Temperatures from (0 to 60) ☌ and for Some Other Aliphatic Carboxylic Acids at (18 or 25) ☌ in Aqueous Potassium Chloride Solutions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
